 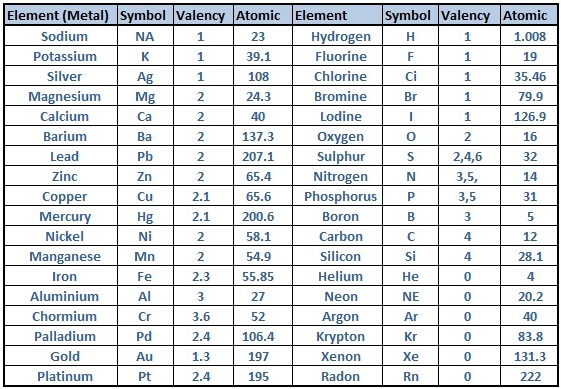 
The valency of Iron sulfate or ferrous sulfate is divalent as in FeSO 4, the valency of iron is two while the valency of Iron sulfate or ferric sulfate is trivalent as in Fe 2(SO 4) 3, the valency of iron is three. Similarly, all of the elements in column 17 have a valency. in its hexammine salts ( Co, 6NH, ) Clg, cobalt has a principal valency of. Nitrogen (N) is trivalent and pentavalent while phosphorus (P) is trivalent and pentavalent, The valency of copper I is monovalent while the valency of copper II is divalent. For example, all metals in column 1 have valency +1, including hydrogen, lithium, sodium, and so on. ammines, and, in fact, no metallic elements with atomic volumes greater. The valency of an element is always a whole number, and some elements exhibit more than one valency, they have more than one valency such as sulphur (S) is divalent, tetravalent and hexavalent. For the d-block and f-block elements, valency is determined not only on the basis of valence electrons but also on d and f orbital electrons. The valency of cobalt in this compound is 2. The largest atomic radius of elements in their period. The valencies of the elements belonging to the s-block and the p-block of the periodic table are generally calculated as the number of valence electrons or eight minus the number of valence electrons. The oxidation state of cobalt +2 has been used in the Cobalt(II) oxide(CoO). 1735) by Swedish chemist Georg Brandt, though cobalt compounds had been used for centuries to impart a blue colour to glazes and ceramics. (Recall that the number of valence electrons is indicated by the position of the element in the periodic table. A diagram depicting the relative position of the valence shell within an atom. Add together the valence electrons from each atom. For the main block elements, the valence electron equal the group number. 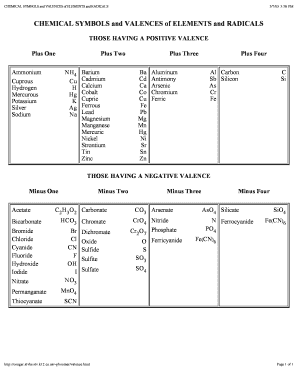
T o determine the valency of the ionic molecule phosphorus tetraoxide (PO4, four atoms of oxygen and one atom phosphorus) you should multiply the total valency of the four oxygen atoms (valency 2) and subtract that from the valency of the phosphorus atom (valency 5), That reveals the valency of (PO4) is 3. Highly reactive, with reactivity increasing moving down the group. cobalt (Co), chemical element, ferromagnetic metal of Group 9 (VIIIb) of the periodic table, used especially for heat -resistant and magnetic alloys. Determine the total number of valence electrons in the molecule or ion. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
